Abstract
Introduction: Treatment targeted at the clonal plasma cells that produce the pathologic immunoglobulin light chain in patients with systemic AL amyloidosis, if effective and timely, can normalize serum free light chains resulting in organ responses and significantly improved overall survival. A third of patients with AL have no response to initial therapy, and over half of those who respond to initial therapy will relapse in 4 years; non-responders and relapsed patients with cardiac involvement are at especially high risk of shortened survival. Single agent daratumumab has been shown to have a response rate of 36% in relapsed refractory multiple myeloma patients (Lokhorst, Plesner et al. 2015). Daratumumab is an option for patients with AL who have marrow plasma cell burden > 10%, consistent with myeloma. Antibody- dependent cellular cytotoxicity is a mechanism of action of daratumumab, and this implicates recognition of the FC portion of the therapeutic antibody by FC receptors on effector cells such as macrophages and NK cells. A single nucleotide polymorphism at codon 158 of the FcγR3A receptor has been shown to affect the therapeutic efficacy of monoclonal antibodies(Mellor, Brown et al. 2013). The aim of this study was to assess hematologic and organ responses to daratumumab in a cohort of AL patients with primary refractory or relapsed clonal plasma cell disease and to test whether FcγR3A genotype influenced response to daratumumab.
Materials and Methods: Patients with biopsy proven AL amyloidosis and organ involvement evaluated at Tufts Medical Center who received daratumumab were identified and evaluated for hematologic and organ responses according to consensus criteria (Comenzo, Reece et al. 2012, Palladini, Hegenbart et al. 2014). FcγR3a genotype for each patient was identified by PCR amplification and sequencing using genomic DNA from blood or marrow mononuclear cells. The translation of 158F or 158V in the alleles of the membrane-proximal extracellular domain was determined by T/G zygosity at nt559 in the gene sequence.
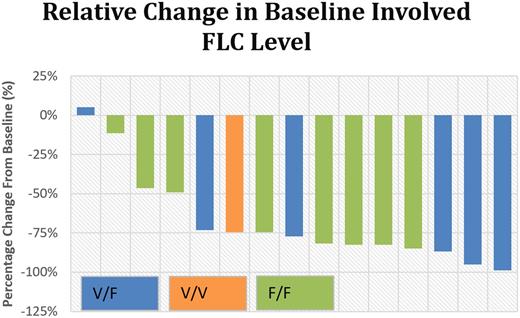
Results: Fifteen patients with AL amyloidosis (λ=10, κ=5) who had primary refractory (no response after at least 2 cycles of initial therapy) (n = 5) or relapsed clonal plasma cell disease (n = 10) received daratumumab at standard doses at a median of 29 months from diagnosis (range, 2-107). Median age at diagnosis was 62 years (47-73) and 60% were male. The median lines of prior therapy were 3 (1-5). Eighty seven percent (n=13) had received bortezomib-based therapy and 53% (n=8) had undergone high-dose melphalan with autologous stem cell transplant. Nine patients had renal involvement (6 with stage 3), and 9 had cardiac involvement with a median NT-proBNP of 2373pg/ml (553-18021). All patients are alive with a median follow-up of 7 months (1-16). Hematologic responses were seen in 87% (CR=5, VPGR=8) with a median time to best response of 71 days. Forty four percent (n=4) had a renal response within an average of 107 days while 55% (n=5) achieved a cardiac response within an average of 149 days. Responses did not correlate with FcγR3A genotype (see figure below). Those with 158V/V or V/F had 3 CR, 3 VGPR and 1 no response (NR) while those with 158F/F had 2 CR, 5 VGPR and 1 NR. The ratio of FcγR3A 158 genotypes in patients with systemic AL amyloidosis has not previously been studied.
Conclusions: Daratumumab achieved rapid hematologic responses in over 85% of patients with primary refractory or relapsed systemic AL amyloidosis with significant organ responses. Daratumumab activity was agnostic to FcγR3A 158 genotypes in this cohort of patients, in whom, of note, the distribution of genotypes is of interest. The basis for daratumumab activity may be linked to other mechanisms such as direct cytotoxicity or to the low fraction of cells in S-phase in AL plasma cell clones (Gertz, Kyle et al. 1989). Daratumumab is a very active agent in AL and its rapid clinical evaluation in patients with newly diagnosed AL is anticipated.
Comenzo: Prothena: Consultancy, Research Funding; Janssen: Consultancy, Research Funding; Karyopharm: Research Funding; Takeda: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal